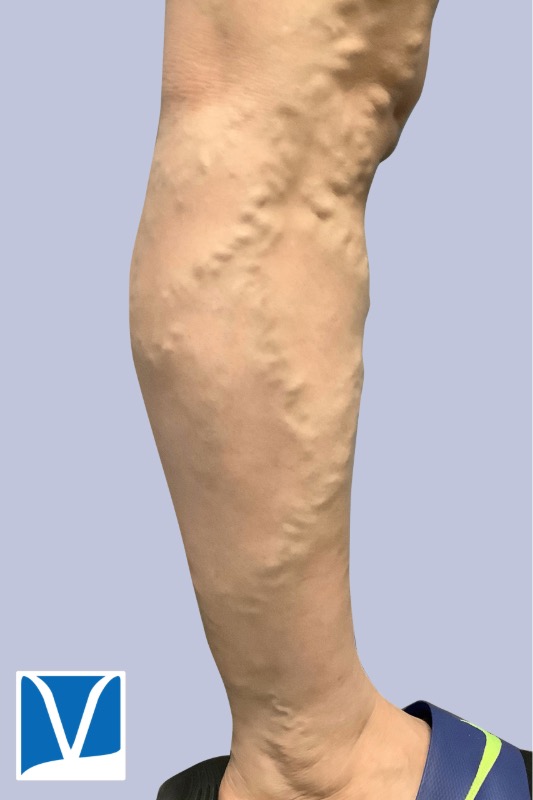
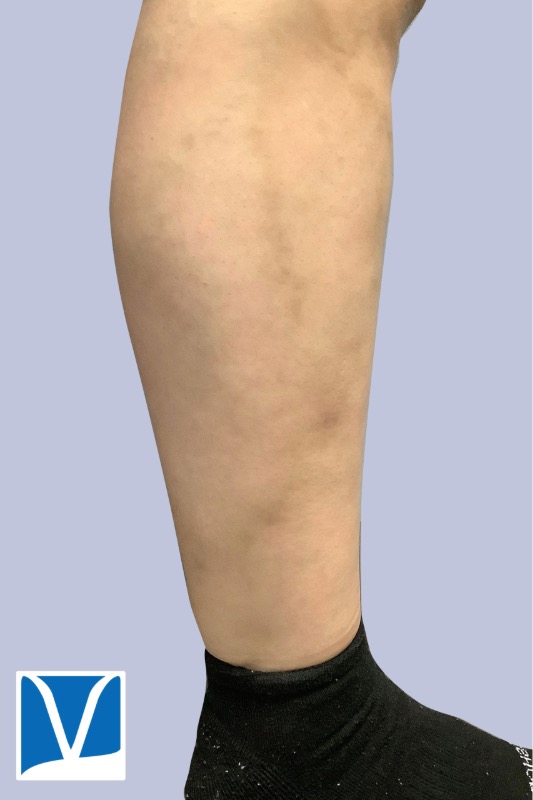
Varithena vein treatment for varicose veins is one of the most exciting developments in sclerotherapy in recent years. Sclerotherapy is commonly used approach to treat small, medium branch varicose veins, as well as spider veins. However the underlying source of these veins is often the greater saphenous vein (GSV) and small saphenous vein (SSV).
Over the years, it has been noted that sclerotherapy to treat the source sapheonous veins that cause most varicose veins is not as effective as treatments such as Closurefast RFA. Furthermore, sclerotherapy was not approved to treat the saphenous veins, nor was it covered by most insurance companies for this purpose.
Sclerotherapy is an injection of an FDA approved medication in either liquid form, or in some cases, as a foam.
Used With Permission | Results May Vary
Common sclerotherapy agents in the united states include Polidocanol and Sotradecol. These agents were originally indicated to treat uncomplicated spider veins (varicose veins ≤1 mm in diameter) and uncomplicated reticular veins (varicose veins 1 to 3 mm in diameter) in the lower extremity. There was not much evidence to support label indications for larger varicose veins (> 3 mm in diameter). Since the saphenous veins are generally far larger than 3 mm, these commonly available sclerotherapy agents were not optimal for the treatment of the saphenous veins that cause most varicose branch veins.
THESE PHOTOS ARE OF AN ACTUAL PATIENT OF OUR PRACTICE WHO HAS PROVIDED CONSENT TO DISPLAY THEIR PICTURES ONLINE. KEEP IN MIND THAT EACH PATIENT IS UNIQUE AND RESULTS MAY NOT OCCUR FOR ALL PATIENTS. PLEASE BOOK A CONSULTATION FOR MORE INFO ON HOW TREATMENTS MAY HELP YOU.
A stronger concentration and optimized foam preparation known to be required to treat the saphenous veins, but this was not commercially available in the United States. Recently, polidocanol endovenous microfoam (PEM) 1%, known as Varithena, was approved for use in the United States by the FDA. Varithena(polidocanol injectable foam) is a stronger concentrated commercially available foam indicated for the treatment of incompetent great saphenous veins, accessory saphenous veins and visible varicosities of the great saphenous vein (GSV) system above and below the knee. This is a specialty formulated foam sclerotherapy that retains its foam confirmation for a longer time, and therefore is more effective at closing the causative truncal veins such as the GSV and SSV.
Varithena is a drug/device combination product that produces engineered polidocanol endovenous microfoa. Varithena vein treatment is delivered from a canister through a syringe into the incompetent vein under ultrasound guidance. Varithena first displaces blood and then the polidocanol chemically ablates the inner lining of the vein wall, causing the vein to close.
In this procedure, a catheter is placed on the inside of the vein, and foam bubbles are injected under ultrasound guidance by the doctor, which causes the veins to spasm, and close off.
This approach is especially best suited for patients with complicated varicose veins, such as those patients who have had prior clots in the veins, making passage of a catheter up the vein more difficult.
Call 1-800-834-6362 to Schedule a Consultation or Use Our
The Varithena Vein Treatment Data
The use of Varithena as a treatment for varicose veins was evaluated in multiple clinical trials where it was shown to be safe and effective. In these studies Varithena vein treatment was shown to improve the symptoms of superficial venous incompetence and the appearance of visible varicosities. We participated in one of these market approval trials, known as VANISH II trial.
Based on the data from these studies, Varithena was approved by the FDA. Furthermore, after evaluation by CMS, Varithena now has insurance coverage for reimbursement when indicated by Medicare and some other insurance companies